I received this letter from Dr. Michael Snyderman, the Director of Clinical Oncology for the WPI, this morning. I am posting it with permission. He is hoping to lend some balance to the discussion. This is what one doctor did with early testing, even before the scientific process had run its course, since he would have been dead before ever starting treatment had he been unable to connect the dots.
Dear Dr. Deckoff-Jones:
It is a shame that the WPI has lost time from having to be on the defensive. I am a believer in MLRVs causing human disease. The supporting data actually goes back to the 1970s. Three different research labs including Robert Gallo’s the co-discoverer of HIV found MLRVs in many different types of lymphoma and leukemia. I believed in “XMRV” enough that when Dr. Mikovits told me that my blood sample was positive, I put myself on AZT and raltegravir for my chronic lymphocytic leukemia and CFS. At the time I started treatment (5/27/10) both were getting worse quickly. I had no treatment for cancer or CFS prior to ARV’s.
I have been a hematologist/oncologist for 40 years. I am tired and am not cured but I am improved at 11 months on treatment. Both the leukemia and the CFS improved coincident with disappearance of infectious virus from my plasma.
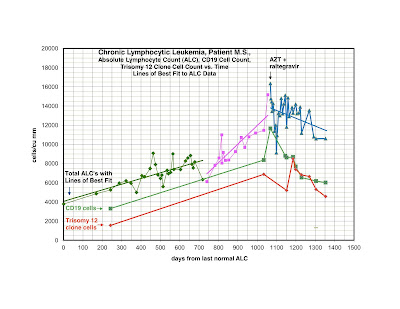
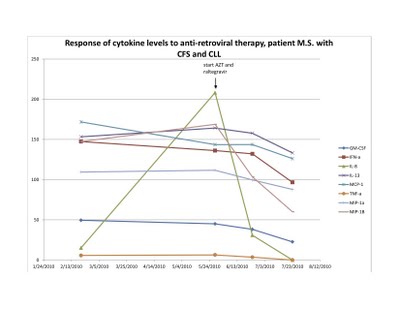
I am plotting total lymphocyte count, total B-cells (CD19) and an unfavorable clone of CLL cells (trisomy12). As you can see, my counts were going up rapidly and now are slowly improving. To my eye it looks convincing that there has been a definite biological change in the leukemia. I also feel somewhat less fatigued and my neuropathic pain is gone. Attached are my lymphocyte count, CD19 (leukemia cells) and trisomy 12 cells (the worst group of cells of my leukemia), as well as my original cytokine profile, vs. 9 months of treatment.
There is no other reasonable explanation than that XMRV, or another retrovirus, was at least making things worse, and subtracting its influence has improved my disorders. This is proof of principle in my opinion. Obviously, an enormous amount of work needs to be done. For example, we don’t have a protease inhibitor, and ARVs are not going to be the only answer. My perspectives are mine, but I believe there is a scientific basis.
I have B-cell CLL with unfavorable prognostic factors: doubling time < 1 year and trisomy 12. I also had the characteristic elevations of cytokines, fatigue and pain seen with CFS. I started AZT and raltegravir May 2010, because I felt things were deteriorating and I had little to lose. More importantly, if I was successful, it could translate into a treatment option for cancer patients that otherwise could not be helped.
The use of ARVs for treatment of cancer is not new; it was used in the 1990s for treatment of HTLV-1 associated lymphoma and did help. I knew then that if I used it on patients I would be vulnerable to censure from my fellow oncologists. I look at the fact that I got CLL as the opportunity of a lifetime to do some good. I am a one person project. I hope sharing my data will encourage people that we will prevail. I will be submitting this data to an online Hematology Oncology journal soon. Some excerpts from the paper are included below.
Michael Snyderman, MD
 |
| Click to enlarge |
 |
| Click to enlarge |
Preview
Review of murine leukemia virus-related viruses and malignancy and outcomes of anti-retroviral drug therapy in a Xenotropic Murine Leukemia Virus Related Virus XMRV positive patient with Chronic Fatigue Syndrome and chronic lymphocytic leukemia.
Abstract
Background: Xenotropic Murine Leukemia Virus-Related Virus (XMRV) was identified in 2006 in some patients with prostate cancer and first isolated in 2009 from most patients with the Chronic Fatigue Syndrome (CFS). Of a cohort of 300 patients with CFS from the 1984 Nevada outbreak, 13 developed B-cell malignancies. The 13 patients were positive for XMRV and a gd T-cell clonal expansion. Causality of XMRV associated CFS and malignancy can be addressed by treatment with anti-retroviral drugs. A patient with both CFS and CLL was available for study. Materials and Methods: Peripheral blood mononuclear cells were isolated. XMRV infection was detected by RT-PCR for gag and env expression; isolation of infectious virus and seroreactivity to XMRV antigens. Cytokine profiles were determined by multiplex analysis of 30 cytokines chemokines and growth factors on a Luminex platform. T-cell clonal expansion was determined by testing for clonal T-cell receptor gamma gene rearrangement (TCRg) by assays available from commercial clinical laboratories. Results: A patient with CFS and B-cell chronic lymphocytic leukemia (CLL) was positive for XMRV and clonal TCRg. He showed improvement in his cytokines, CFS symptoms and hematological parameters simultaneous with disappearance of infectious XMRV in plasma after treatment with AZT and raltegravir. Conclusions: Improvement of both the CFS and CLL simultaneous with disappearance of infectious XMRV from plasma after anti-retroviral therapy suggests that XMRV was etiological for both. Anti-retroviral therapy might have a positive impact on CFS and XMRV associated malignancy in other patients.
Results
A patient with CFS and B-cell CLL tested positive for XMRV in both plasma and leukemia cells. His virus was sequenced and was closely related to the previously sequenced XMRV strains in our laboratory. He had antibodies to XMRV proteins. He was also positive for a clonal TCRg by both quantitative and qualitative assays. He had unfavorable prognostic factors including a trisomy 12 clone and a doubling time of less than one year and had no prior therapy. He started treatment with AZT and raltegravir 571 days after diagnosis. By day 56 of treatment, infectious XMRV was no longer detectable in plasma and his cytokine levels had improved. This coincided with improvement in symptoms of CFS which included fatigue, difficulty in concentration and neuropathic pain. He continues on treatment for eight months with good tolerance of drugs and works full time. His previously increasing absolute lymphocyte count (ALC), CD 19 cells and trisomy 12 cells are trending downward. At start of treatment, his ALC was 16,348/cu mm and CD 19 cells 11,658/cu mm up from 3,303 at diagnosis. His trisomy 12 cells peaked at 8,490/cu mm day 117 of treatment up from 1,550 at diagnosis. After 285 days of treatment his ALC was down to 10,600/cu mm, CD 19 cells down to 6,015 and trisomy 12 down to 4,558.
Discussion
We have previously reported that most CFS patients are positive for XMRV if multiple detection methods are used. The development of lymphoid malignancies in 13 of 300 CFS patients suggests that CFS patients are at a several hundred fold increased risk for malignancy compared to generally quoted incidences in the general population. Of these 13 patients, all that were tested were positive for both XMRV and a clonal gd T-cell expansion. The greatly increased risk for B-cell malignancy in an XMRV infected population may be due to infection of the B-cell line by XMRV. Cancers have mutated genes and changes in gene expression that could make them permissive to infection by retroviruses. Retroviruses have been thought to cause cancer by insertional mutagenisis. This mechanism requires that the retrovirus proviral DNA be integrated into host cell DNA next to a proto-oncogene thereby inducing activation of the proto-oncogene. A more important mechanism with XMRV may be the ability of viral proteins to change gene expression. Several independent groups have found that shortly after a permissive cell line is infected with XMRV, multiple genes are expressed and that these genes could be involved in production of the malignant phenotype. Spadafaro has shown that reverse transcriptase can cause gene activation and lead to the malignant phenotype. In some cancers env and gag may also be important in malignant transformation.
A complementary hypothesis is that T-cells are also infected by XMRV resulting in a clonal T-cell expansion. The clonal T-cells produce elevated cytokine levels which may be partially responsible for the CFS. Furthermore these cytokines may have a paracrine activity that stimulates a simultaneous neoplasm to behave in a more aggressive fashion.
One objection to considering XMRV a pathogenic virus is that there was previously no explanation as how a murine leukemia virus could have entered the human population. However, early vaccines were prepared by passaging human virus through mice for the purposes of viral isolation and for attenuation. This would have allowed for contamination of vaccines with murine leukemia viruses. The original Yellow Fever Vaccine was made in the early 1930’s by culturing the virus in mouse cerebral tissue. Some patients received both the Yellow Fever virus and infected mouse cerebral tissue.
The YF17D strain was used to immunize over 400 million people world-wide over the next 65 years. It was the policy of the U.S. Armed Forces to vaccinate service men for Yellow Fever from the 1940’s through the 1970’s. The polio vaccination trials in the United States started in 1952. The polio virus was passaged through mice, cotton rats and primates to achieve attenuation. Although the Salk vaccine was treated with formalin, many patients received the live oral attenuated polio virus. Indeed, the patient studied here, received the live oral polio vaccine in the early 1950s, fifteen years later developed symptoms of CFS and fifty-five years later developed CLL. He also received the Yellow Fever vaccine in the early 1970’s on entering the Armed Services.
In summary, a new patient with both CFS and B-cell CLL was identified. Both his leukemia cells and plasma were positive for XMRV and he had antibodies to viral proteins. He also was positive for a clonal TCRg and had elevated cytokine levels. With anti-retroviral therapy he showed improvement in his cytokine levels, CFS symptoms and hematological parameters simultaneous with the disappearance of infectious XMRV from his plasma. Presumably his improvement was related to the anti-retroviral effects of treatment.
Alternative explanations for the therapeutic effect of his anti-retroviral therapy have been offered. One of these is “selective toxicity” which has never before been seen with the hundreds of agents use as cancer therapeutics and seems an unlikely explanation for his improvement. Anti-telemorase activity of the AZT has also been considered, but the rapid response to treatment does not fit the kinetics of depletion of telomers.
There is nothing unique about this patient’s clinical presentation to suggest that his case is any way unrepresentative. His response to anti-retroviral therapy suggests that XMRV was etiological for both his CFS and CLL and that anti-retroviral therapy might help other patients with CFS and XMRV associated malignancy. Many more patients need to be studied. Ultimately questions that should be answered are what neoplasms are associated with XMRV, will existing anti-retrovirals have activity in these neoplasms and what would be the optimal combination of anti-retrovirals.
Did you like this? Share it:



>Thanks! Great to hear advances and progress.
>Fantastic!!!!
>Thank you Dr. Jamie for posting Dr. Snyderman's stunning account of his illnesses and how they related to XMRV. This is compelling proof of XMRV causation or effect in CFS and his CLL.
It will be interesting to see the response to the article once it is online.
APPROPRIATE TESTING NOW! CLINICAL TRIALS NOW!
>Thanks so much for posting. It gives hope. May I ask if Dr. Snyderman had any other cancer treatment other than retroviral therapy?
>Thank you, Dr. Snyderman, for doing all this for sufferers, and thank you, Dr. Deckoff-Jones, for posting it. I am grateful.
Patricia Carter
>Thank you. For me this is another validation that we are on course. Marilyn Bloomer
>I read recently that Isentress (Raltegravir) has the ability to address herpesviruses such as HHV6.
Is there any possibility that the reason that people with CFS improve on arv's is because their herpesviruses and not a retrovirus are being addressed?
Some herpes antivirals (Valcyte, Valtrex, etc.) have been known to move the illness in a few cases too.
>Anonymous 1:!6, Dr. Snyderman had no other cancer treatment prior to arv's.
Jamie
>Please thank Dr Snyderman for his invaluable contribution to the understanding of ME. He is most unfortunate to be as well placed as he is to make this contribution but millions of people will benefit from his "project of one".
I wish him continued improving health!
>Posts like this are extremely important. Thank you very much to both of you. We have had little news lately and we are getting impatient for more.
We all need to keep the goal for all of us in mind when we do things and both of you are doing that.
Are the green, pink, and blue lines different regions of fit for the same measurement?
Also, after you subtract signs and symptoms that are CLL but not the other disease do you end up with those 3 symptoms or something like CCC?
>Samuel,
Linear lines of best ("trend lines") fit were used for total lymphocyte counts only. Each period is different. Early on the rise in the lymphocyte count was favorable, over 1 year to double. In the second phase things were worse and the doubling time was unfavorable at less than a year. The third phase is with treatment. Presumably with treatment the CLL will revert to a more favorable prognosis.
Michael Snyderman
>Thank you for posting this Dr Deckoff-Jones and thank you Dr Snyderman for sharing this with us.I am sorry for your illness but this gives me some hope.I hope you continue to do better.
>And why would then would leaving the XMRV studies be a good idea as was stated in the recent State of Knowledge conference??? I'm not a scientist but that seems to make no rational, scientific sense at all.
>After a quick web search, I'm wondering even more if an alternative explanation for Dr. Snyderman's experience is that the Raltegravir addressed EBV, and that retroviruses have nothing to do with it.
e.g.
http://www.aidsmeds.com/articles/hiv_herpes_Isentress_1667_19150.shtml
http://www.infectagentscancer.com/content/5/1/22
A question, of course, is, why is EBV activated and causing problems?
An immunocompetent host should be able to keep EBV under control. But there are other reasons for immune systems to be compromised other than retroviruses.
Dr. Snyderman says there can be no reasonable alternative explanation to the idea that a retrovirus caused his results.
Can this one be ruled out?
>Thank you Dr Snyderman and Dr Deckoff-Jones for your testimonies to the effectiveness of ARV treatment of Myalgic Encephalomyelitis (the true name of the epidemic disease covered up by CFS). This is compelling evidence of the cure confirming the cause, and your case histories will add to the pressure that clinical trials should be funded as soon as possible.
For Anonymous regarding could it just be EBV, have you watched the 1996 Primetime Live report on CFS? If you had you would know that Cheney and Peterson had brain scans of CFS patients that were identical to AIDS patients, which prompted them to find the low NK cell dysfunction and Elaine de Freitas to find that retrovirus, which the CDC squashed. It could be one of the variants of XMRV as this story is still unfolding or it could be another retrovirus causing similar symptoms.
CFS and the CDC's Failure to Respond: Primetime Live (1996) http://www.youtube.com/watch?v=AW0x9_Q8qbo
>Thanks very much for sharing this amazing information. I am grateful for Dr. Synderman sharing this information publicly. I hope that he will send this information specifically to some of the scientists working on XMRV/MLV studies like Dr. Harvey Alter, etc.
>Thanks, Dr. Snyderman. Nice documenting and reporting. Full-time work is a great milestone to have achieved. Best wishes for continued success.
I have to wonder if we have to broaden the anti-retroviral treatments because of Coffin's suggestion that Xmrv is not a primary cause for ME/CFS. Maybe it's another MLV/HGRV that is main-causal and needs treatment.
Miles to go, yet. ..And oh yeah, I remember that sugar cube, and I think later, a sweet drink a few months before getting quite ill.
>cud a retro virus be resposbale for diseases like trisonmoy 12, turner syndrome, fragile-x, and other 'genetic' diseases?
I would be very curious to know if dr Snyderman has a simian line on either of his hands?
google simian line for pictures.
again thanks to Jamie
and a big thank you to Dr.Snyderman for sharing his information with us.
fly
>Thank you Dr. Synderman. Your courage inspires. I cannot help but think there could be fallout towards your professional reputation. Given that possibility, I direct my prayers this Easter Weekend to you and yours, and also pray you continue to improve.
Thank you for your courage under fire!
~ katie weatherford
>Thanks so much for posting, and thank you Dr Snyderman, I do so hope that it further treament your health imporves further. And I hope you will be sending this to Dr MAangen and Dr Collins of the NIH.
>Thanks Dr. Snyderman. I hope your health continues to improve. You will be in my prayers.
Scott
>Dr. Snyderman rocks! So glad he is doing this excellent work!! Thank you!
>Anonymous, did you even bother to read the literature you provided:
http://www.infectagentscancer.com/content/5/1/22
"In CLL, evidence for EBV infection was obtained only in a subset of cases and, notably, only in a variable fraction of neoplastic cells. These findings contrast with the constant presence of EBV genome in all tumor cells of malignancies pathogenically linked to the virus and argue against a causal role for EBV in the early phases of CLL development. Although a partial loss of viral genomes can not be formally excluded in these cases, this possibility appears however unlikely. Moreover, the fact that viral products necessary to sustain the transformed phenotype may be expressed only by a fraction of tumor cells further weakens the hypothesis of a pathogenic involvement of EBV in this tumor. Although further studies are required to elucidate this complex and still debated issue, available evidence briefly reviewed herein is consistent with a POSSIBLE role of EBV infection as a secondary event occurring after cell transformation and affecting a subpopulation of CLL cells in which the virus may promote the evolution to a more malignant phenotype. The possible prognostic role of EBV-driven production of factors able to promote CLL cell growth, angiogenesis and to inhibit tumor-specific immune responses deserves to be carefully investigated by prospective studies. If such progression mechanisms will be conclusively demonstrated, the use of antagonists or inhibitors of molecules produced by EBV-infected cells could represent a potentially attractive therapeutic approach."
As they say, it's the retrovirus, stupid. They found it 20 years ago, only the Centers for Drug Control botched it in my opinion and have continually punted the political football ever since.
http://www.ncbi.nlm.nih.gov/pubmed/1672770
I developed AIDS-related complex a mere two years after catching the flu that never went away in 1992, despite testing negative for HIV but didn't lose the capacity to exercise, much like Dr. Jones herself, until some 7 years in, wherein I developed NMH and diastolic issues (I always suffered from the extreme fatigue, tender lymph nodes, chronic headache and intestinal distress). As EBV is ubiquitous in the population, how is it that I infected my girlfriend and now wife, who, quite unlike my early experience, was rendered couchbound immediately upon infection? Did I somehow trigger her own EBV through bad karma (she had mono as a teenager)?
My brother and sister both suffer varying degrees of debilitation. My father developed a meningitis-type illness that almost took his life and "locked" one of his eyes that needed to be surgically unlocked. He has since developed colon cancer that spread to the liver. I suppose this is all just attributable to an extremely virulent form of one of the most well-studied viruses of all time.
Is EBV a neurotropic virus capable of microglial activation and increased cytokine counts of a particular type in the spinal CFS proteome?
EBV may play a role, but only in piling on in an already compromised host. That isn't to say that treating it wouldn't potentially mitigate the illness.
– Former microbiologist.
>Please post this. I'm begging you this time. I have had CFS for more than 9 years, during which time I spent every spare moment researching the subject. I finally came across a website claiming to cure, and offering a money back guarantee. It is a form of molecular and cellular agents. PLEASE just looke at their website -www.digestacure.com
I have no vested interest in this company, except the fact that they gave me my life back. I am free of CFS. Sunny Lindsay sprince@xmission.com.
>OK, I did just have a quick look at their website. I found that digestacure is owned by Pristine Nutraceuticals, LLC who, despite being in business for less than a year, have already earned an "F" grade from the Better Business Bureau:
http://www.bbb.org/south-east-florida/business-reviews/vitamins-and-food-supplements-retail/pristine-nutraceuticals-in-fort-lauderdale-fl-92020422
>To Former Microbiologist –
This article states that EBV is not causal in CLL. And I think that just about everyone agrees that EBV is not causal in CFS.
Apparently something about CFS sufferers’ systems — and apparently, CLL sufferers’ systems — allows EBV and other pathogens to proliferate.
As I said, an immunocompetent host should be able to keep EBV under control. Something is compromising CFS and CLL sufferers' immune systems.
One hypothesis here is the thing that is preventing these people from keeping the EBV under control is a retrovirus.
But this is just one possibility. There are other things that could be causing people’s systems to be immunocompromised and allowing EBV to reactivate.
The reason that I brought up this paper is because Dr. Snyderman stated the following: “There is no other reasonable explanation than that XMRV, or another retrovirus, was at least making things worse, and subtracting its influence has improved my disorders.”
This seems to be an open invitation to figure out alternative explanations.
The alternative explanation that I posed is that EBV (reactivated for some unknown reason that has nothing to do with retroviruses) was serving as a progression factor in Dr. Snyderman’s illness; that the Raltegravir he took got the EBV under control; and that his illness got better as a result of addressing the EBV.
Dr. Snyderman does not report getting well. He just improved. That would be consistent with the idea of reversing a progression factor.
The same thing could have happened with Dr. Deckoff-Jones and her daughter. In the past, she has used the concept that the drugs “move the illness” as evidence that XMRV is present and causing at least some symptoms.
Even if we assume that their improvements were due to the drugs, that doesn’t mean that the drugs addressed a retrovirus or that there was a retrovirus present at all.
Again, maybe they both had active EBV infections (resulting from being immunocompromised for some reason other than having a retrovirus), and the Raltegravir helped to get those infections under control — thus allowing them to improve (as you’d expect when addressing a serious infection) but not get close to wellness.
Obviously, EBV is not causing people to have CFS.
The idea that a retrovirus is at least partly causal makes sense, based on what we know about this illness, about retroviruses in general, and about XMRV.
But the idea that a retrovirus makes sense is not the same thing as saying that there actually is a retrovirus present or causing problems. Maybe something totally different is causing sufferers' systems to be immunocompromised and resulting in the other problems that you mention.
Until all the alternative explanations are ruled out, there’s no way to know.
>Nicely explained, former microbiologist.
>"CFS" itself came into existence because it was kind of silly for us to have to say "The illness is called CEBV Syndrome, but we already know that EBV is not the cause".
The CDC grudgingly admitted that this made them look pretty dumb, and came up with "CFS".
Except, of course, they took the credit for discovering that EBV didn't fit the facts, even when the real story was in all the newspapers.
>I am XMRV+ with ME now on ARV's and improving slightly. My wife is now diagnosed with acute myeloid leukemia and having chemo . Is there anything to stop her being tested for Xmrv and if positive taking say AZT and ten.?
>"The supporting data actually goes back to the 1970s. Three different research labs including Robert Gallo's the co-discoverer of HIV found MLRVs in many different types of lymphoma and leukemia."
….Would it please be possible for you to provide us with any information on the evidence that Dr Gallo & other labs found these retroviruses back in the 1970s?
Are there any papers published on this?
Is Dr Gallo going to make a statement about this? If he knew they were there, why hasn't he said anything?
Please can you give us more information.
A London scientist.
>What is the difference between MLUV's and MLRV's?